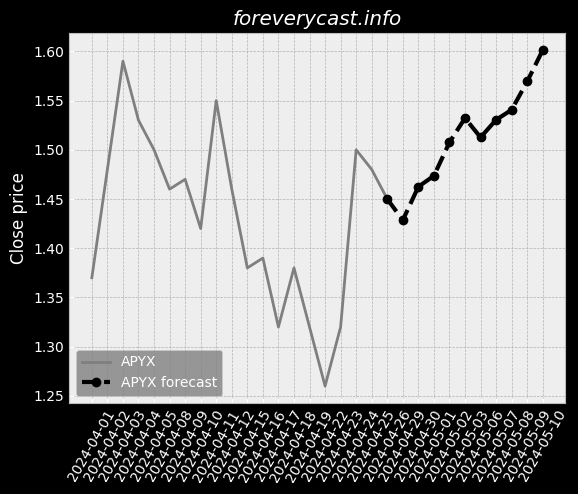
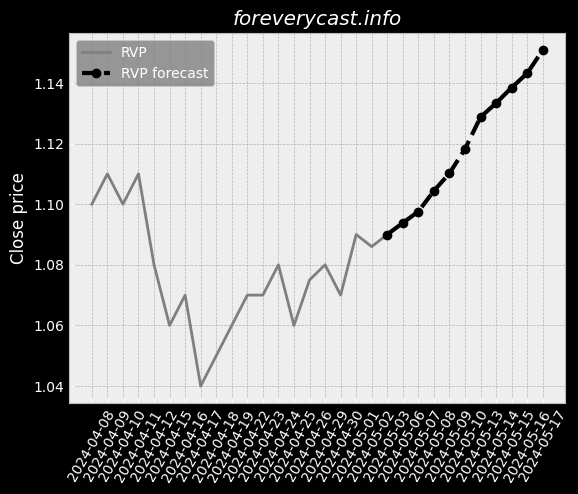
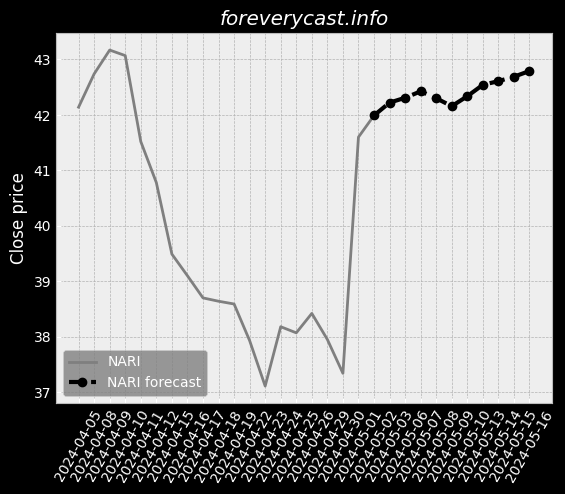
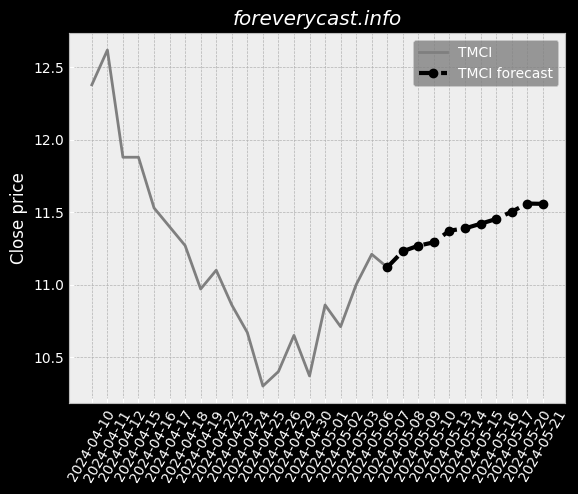
To date we have had no issues in our facility. & The second study was completed in June 2017 and involved 46 patients. of clear liquids or two doses of 7.5 oz. We received a general approval for such transfer. We believe our relations with our employees are good. 9679; Class III devices present the highest risk. Prior approval is obtained through submission of a PMA supplement. The collection of such data may be required as a condition of PMA approval. We cannot assure you, however, that we will be able to achieve any of the foregoing. 25 & & Our Pure-Vu System may in the future be subject to product recalls. We are a medical technology company with a limited operating history. & The Pure-Vu System may not be accepted by physicians and patients. & As of December 31, 2020, we had 24 full time employees. We may pursue acquisitions of businesses and assets. We may face risks related to health epidemics or outbreaks of communicable diseases. We are an “emerging growth company” as defined in the JOBS Act. There is little binding case law in Israel that addresses the matters described above.