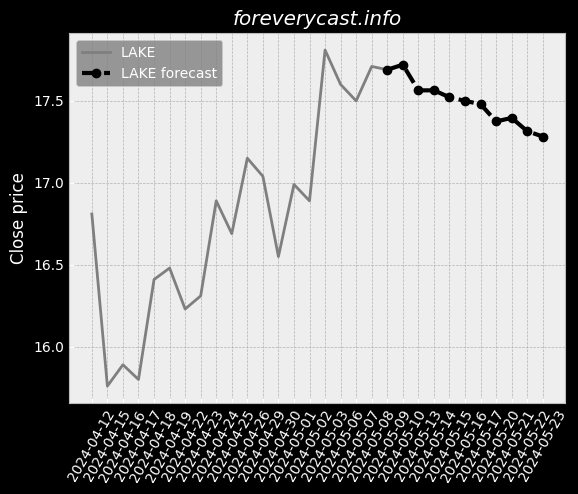
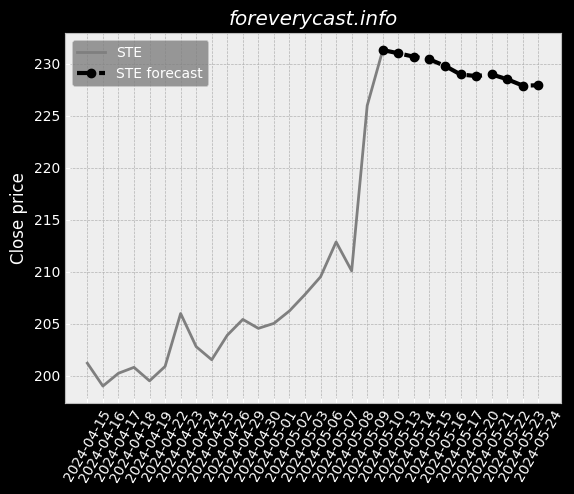
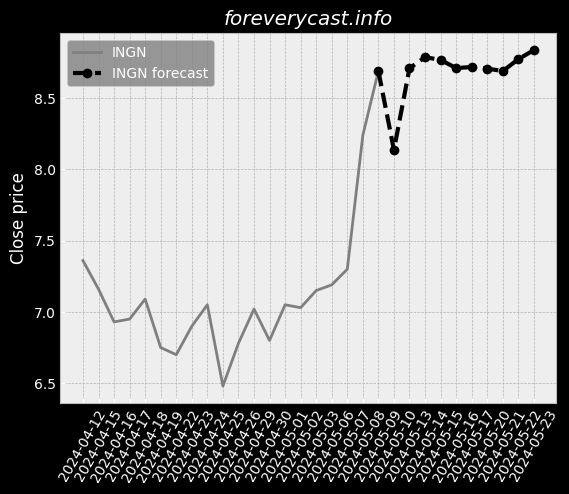
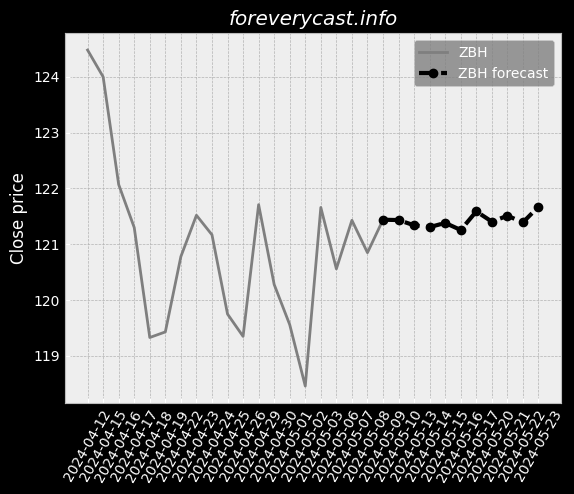
The miraDry segment focuses on sales of the miraDry System and bioTips. Plastic Surgeons are thought leaders in the medical aesthetics industry. miraDry Strong clinical trial outcomes. Broaden our product portfolio and launch new products and services. However, we do not obtain a material portion of our revenue from our service contracts. miraDry We have implemented targeted marketing and practice support programs. The FDA has inspected both the Company and our suppliers. To a lesser extent, we indirectly compete with antiperspirants. 24 The FDA may impose additional post-market requirements. Noncompliance with the federal AKS could result in the penalties set forth above. Federal Criminal False Claims Laws. Medical data is considered sensitive data that merits stronger safeguards. Physician Payments Sunshine Act. There are risks related to our intellectual property rights. For the year ended December 31, 2020, our net loss was $89.9 million. We have limited manufacturing experience. At this point, the duration and extent of such impact is uncertain. Litigation may be necessary to defend against these claims. Any litigation or the threat thereof may adversely affect our ability to hire employees. Clinical data is ongoing for our second or "new enrollment" post-approval study.