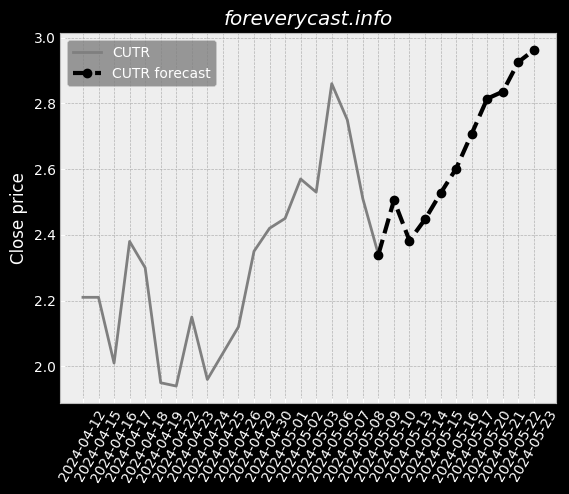
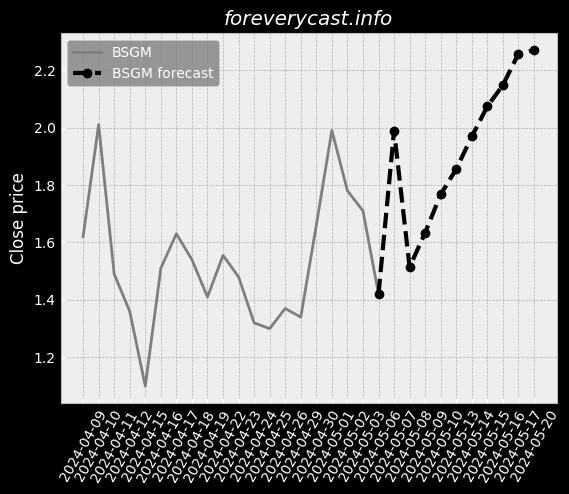
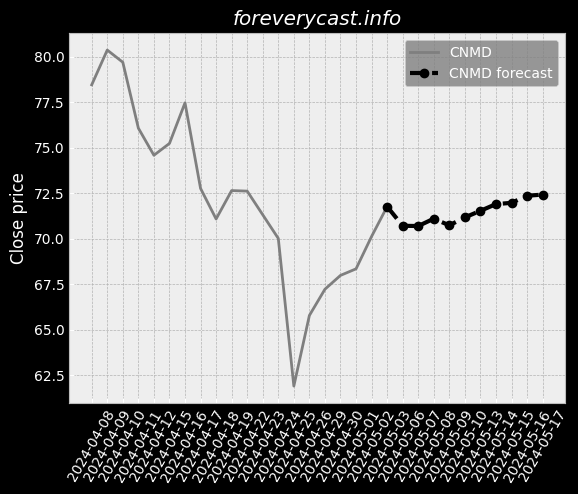
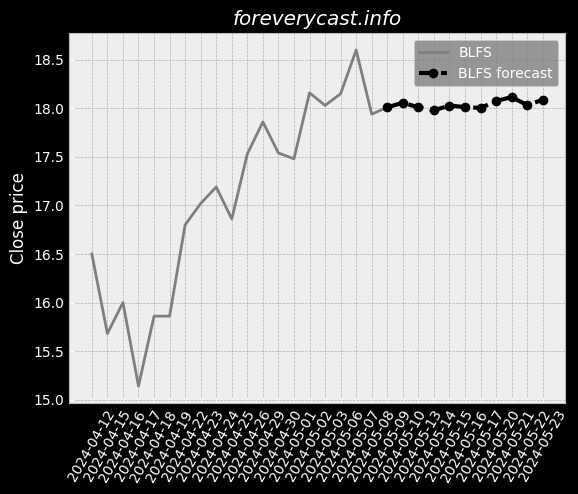
Our mission is to improve patient outcomes and reduce the cost of patient care. In 2013, we also began selling our pulse oximetry products in the consumer market. A low hemoglobin status is a condition called anemia. SpHb® is available as a continuous or a spot-check measurement. Elevated CO levels may cause severe neurological damage, permanent heart damage or death. The lower the number, the less variability there is in Pi over a respiratory cycle. In 2020, we launched four additional products for the consumer market. Masimo rainbow SET® MX Circuit Boards. Radius-7® with Root® has received both CE Mark and FDA 510(k) clearance. Rad-8® is an affordable, low-cost design with a streamlined feature set. RAS-45 has received FDA 510(k) clearance and the CE Mark. Other Agreements with Cercacor. The MDR will apply to the medical devices we commercialize in the EU after May 26, 2021. Instead of a CE mark, medical devices marketed in Great Britain must bear a UKCA mark. Several states have similar requirements. These negotiated prices are made available to a GPO's members. We believe that other entities are manufacturing and selling counterfeit Masimo sensors. The standard for recalling deficient products may be different in foreign jurisdictions. We may also experience limitations in employee resources. If these risks materialize, our stock price could be materially adversely affected. We do not specifically insure against environmental liabilities.