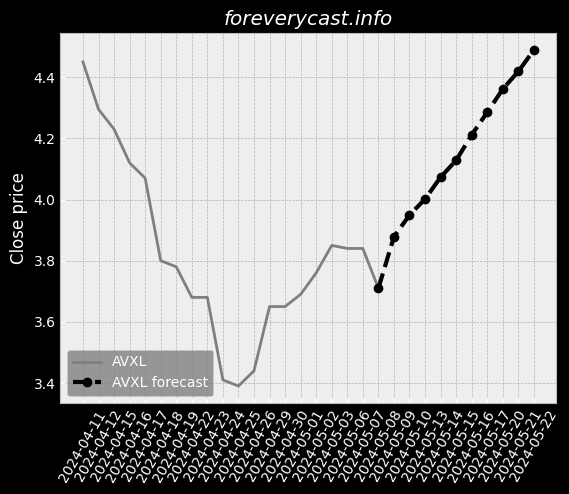
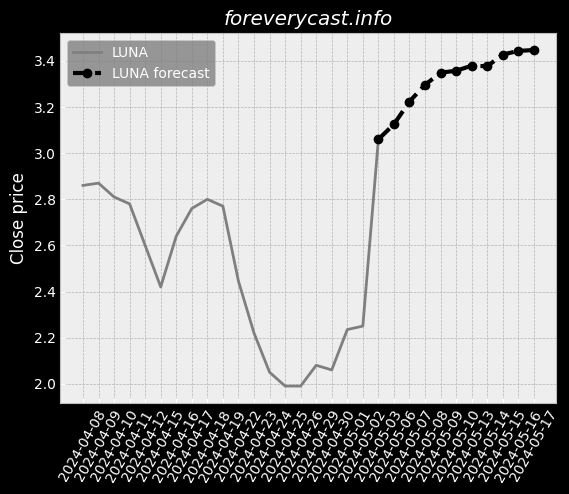
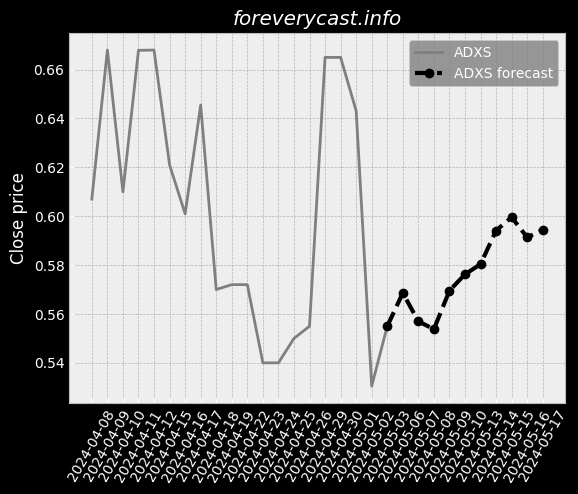
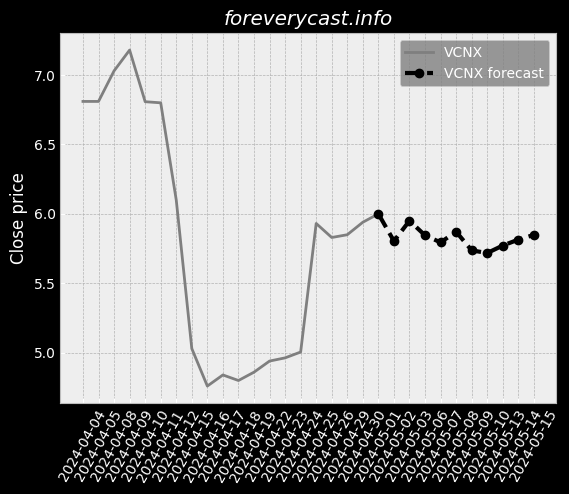
Our current products and services focus on screening tests and precision oncology tests. Covid-19 TestingIn late March 2020, we began providing COVID-19 testing. We currently manufacture our Cologuard test at two facilities in Madison, Wisconsin. These competitors may have broader product lines and greater name recognition than we do. Federal and State Billing and Fraud and Abuse LawsAnti-fraud Laws/Overpayments. From time to time we have identified overpayments and made refunds to government payers. Available InformationWe were incorporated in the State of Delaware on February 10, 1995. This discussion highlights some of the risks that may affect future operating results. These are the risks and uncertainties we believe are most important for you to consider. We cannot be certain that we will successfully address these risks. Our headquarters are also located in Madison, Wisconsin. We undertake acquisition activities from time to time. In late March 2020, we began providing COVID-19 testing. IVDs like our Cologuard test are regulated as medical devices by the FDA. Most states also have similar regulatory and enforcement authority for devices. We may seek to partner with others to assist us with any or all of these functions. Generally, the agreements under which we gain access to human samples are non-exclusive. We may not be able to successfully establish and maintain such intellectual property. As of December 31, 2020, we had $1.84 billion in combined cash and marketable securities.